 It is also sometimes used to refer to the equilibrium states shown on a phase diagram, described in terms of state variables such as pressure and temperature and demarcated by phase boundaries. The term phase is sometimes used as a synonym for state of matter, but there can be several immiscible phases of the same state of matter (as where oil and water separate into distinct phases, both in the liquid state). : 86 : 3 Examples of physical properties include density, index of refraction, magnetization and chemical composition. More precisely, a phase is a region of space (a thermodynamic system), throughout which all physical properties of a material are essentially uniform. 
The glass of the jar is another separate phase. In a system consisting of ice and water in a glass jar, the ice cubes are one phase, the water is a second phase, and the humid air is a third phase over the ice and water. 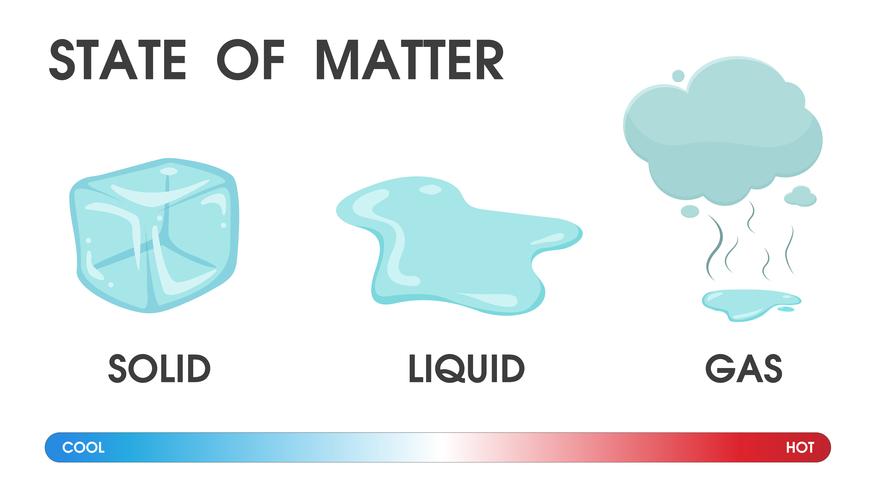
To learn more about salt and ice, check out these snow and ice experiments.In the physical sciences, a phase is a region of material that is chemically uniform, physically distinct, and (often) mechanically separable. Salt lowers the freezing point of water and is often used to melt dangerous ice off of roads and sidewalks in the winter. When that happens, the water is said to be super-cooled.Ĭan you think of any other ways to keep water from freezing when temperatures are below freezing? When there isn’t one, water can reach a temperature below the freezing point without turning into ice. Normally, when water reaches 32° F it begins to freeze.Īs you learned in the super-cooled water experiment, water needs a nucleation site, or a spot for the first ice crystals to form. Those water droplets could then freeze into (solid) ice.Įven with all of these state changes, it is important to remember that the substance stays the same-it is still water, which consists of two hydrogen atoms and one oxygen atom.Ĭhanging states of matter are only physical changes the chemical properties of the matter stays the same regardless of its physical state! When steam comes into contact with cool air (which reduces energy), it can condense back into water droplets (liquid again). (Steam is the gas state of water, and is also called water vapor.) When it begins to boil, some of the water turns into steam. Water is usually a liquid, but when it reaches to 32° Fahrenheit (F), it freezes into ice. 
Water is unique because the properties of water allow it to exist in all three states of matter! 
Note that not all substances can change states just by adding or removing heat-sometimes other physical changes, such as increased pressure, are needed to change the state of a substance. However, if you use heat (i.e., add energy) to melt peanut butter, its state will change and it will flow like a liquid! It acts more like a solid even though it is very soft. Matter can change from one state to another when physical conditions change when energy, such as heat, is added or removed, a substance can change from a solid to a liquid, or from a liquid to a gas.įor example, peanut butter does not flow like a liquid. Go here to see what the molecules of substances look like as a solid, liquid, and gas. The molecules in a gas are even farther apart than in a liquid and move freely with no pattern at all. The movement is what makes a liquid fluid (or pourable) and take the shape of a container it is in. In a liquid, molecules are farther apart, can move around, and are not arranged in a pattern. When an object is a solid, its molecules are arranged in a pattern and can’t move around much. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
